|
4/8/2024 0 Comments Dalton atomic theory
Permission is granted to reproduce for non-commercial educational purposes. This exercise asks whether the atomic weights Dalton assigned to hydrogen, nitrogen, and oxygen were consistent with his assumptions and with the chemical analyses available to him.įurther information: A detailed summary of key primary literature on multiple proportions, the atomic hypothesis, and atomic weights, including some quantitative treatment of data may be found in Leonard Nash, "The Atomic-Molecular Theory," in James Bryant Conant, ed., Harvard Case Histories in Experimental Science, vol. Some of Dalton's contemporaries saw these assumptions as arbitrary. The laws of definite proportions and multiple. 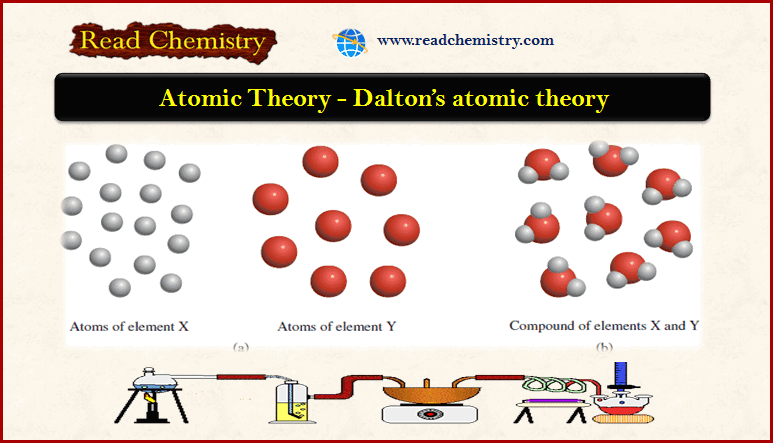
One assumption was that if two elements formed only one compound, that compound consisted of a single atom of each element (for example, water would be HO and ammonia NH). His theory pictured chemical compounds much as we do today, as atoms of different elements bound together. The table was based on chemical analyses and on certain assumptions about what we would call molecular formulas. Using evidence from the previous century of chemical experimentation, Dalton argued that the basic chemical elements are made of atoms, and he started to. When Dalton set out his atomic hypothesis, he included a table of atomic weights relative to hydrogen = 1. He also seems to have believed that they were indestructible. Dalton believed that atoms of the same element were identical. As valuable and fruitful as Dalton's work certainly was, it was mistaken in several details. The fruitfulness of Dalton's theory can be seen in the research on combining masses and relative masses of the elements and their compounds that followed it. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. 1 ), is a fundamental concept that states that all elements are composed of atoms. Dalton focused on properties of atoms that were amenable to empirical investigation, namely their relative masses. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 4.2. The laws of definite proportions and multiple proportions follow naturally from such an atomistic view of chemical combination. His theory pictured chemical compounds much as we do today, as atoms of different elements bound together. Notes: John Dalton (1766-1844) is best known for formulating an atomic theory that proved to be enormously fruitful, even though it turned out to be incorrect in several important details. 
Reference: John Dalton, A New System of Chemical Philosophy, Part I, (Manchester, 1808, 1810) Dalton atomic weights: teaching notes Dalton atomic weights Content: composition, formulas, stoichiometry A New View of the Origin of Daltons Atomic Theory : A Contribution to Chemical History, Together With Letters and Documents Concerning the Life and Labours of.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
